20 POINTS AND GIVING BRAINLIEST
The following reaction shows the products when sulfuric a...

Chemistry, 22.05.2020 01:05 baileyflemingde
20 POINTS AND GIVING BRAINLIEST
The following reaction shows the products when sulfuric acid and aluminum hydroxide react.
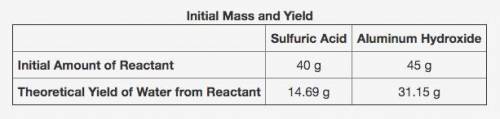
2Al(OH)3 + 3H2SO4 → Al2(SO4)3 + 6H2O
The table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. (See attached image)
What is the approximate amount of the leftover reactant?
A) 20.89 g of sulfuric acid
B) 22.44 g of sulfuric acid
C) 21.22 g of aluminum hydroxide
D) 23.78 g of aluminum hydroxide

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 19:30
What is the mass of oxygen gas is consumed in a reaction that produces 4.60mol so2
Answers: 3

Chemistry, 23.06.2019 06:00
Each step in the following process has a yield of 70% ch4 + 4cl2 yield ccl4 +4hcl ccl4 + 2hf yield ccl2f2 + 2hcl of 4.50 mole ch4 reacts what is the total amount of hcl produced
Answers: 3

Chemistry, 23.06.2019 06:10
How much would the freezing point of water decrease if 4 mol of nacl were added to 1 kg of water (kf=1.86 degrees c/(mol/kg) for water and i=2 for nacl a- 7.44 degrees c b- 14.88 c 3.72 d 1.86
Answers: 1
You know the right answer?
Questions

English, 11.12.2020 03:10

Mathematics, 11.12.2020 03:10



Mathematics, 11.12.2020 03:10


Chemistry, 11.12.2020 03:10

Mathematics, 11.12.2020 03:10

Mathematics, 11.12.2020 03:10

Mathematics, 11.12.2020 03:10


History, 11.12.2020 03:10

Health, 11.12.2020 03:10

Mathematics, 11.12.2020 03:10

English, 11.12.2020 03:10

Mathematics, 11.12.2020 03:10

Mathematics, 11.12.2020 03:10

Mathematics, 11.12.2020 03:10

Mathematics, 11.12.2020 03:10

Mathematics, 11.12.2020 03:10



