
Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 22.06.2019 15:00
‘which reaction would most likely require the use of an inert electrode?
Answers: 1

Chemistry, 22.06.2019 22:30
What is a number added in front of a formula in order to balance the equation
Answers: 1

Chemistry, 23.06.2019 00:30
•hydration •dissociation •dissolving which one goes to which
Answers: 1
You know the right answer?
What is the concentration of each ion in 0.250 M Na2SO4?...
Questions

Computers and Technology, 07.03.2020 05:12




Mathematics, 07.03.2020 05:12



Chemistry, 07.03.2020 05:12







Mathematics, 07.03.2020 05:12





Mathematics, 07.03.2020 05:12

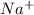 is 0.500 M and
is 0.500 M and 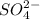 is 0.250 M in 0.250 M
is 0.250 M in 0.250 M 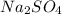
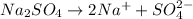
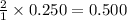 moles of
moles of 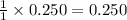 moles of
moles of 

