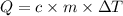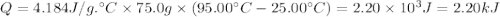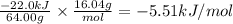Chemistry, 07.05.2020 12:58 dinadenoirefan
What is the MOLAR heat of combustion of methane(CH₄) if 64.00g of methane are burned to heat 75.0 ml of water from 25.00°C to 95.00°C?(Water heat capacity-4.184 J/g°C)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1

Chemistry, 22.06.2019 21:20
40dm3 of gas at 760 torr are heated from 5°c to 50°c what is the new volume
Answers: 3

Chemistry, 23.06.2019 00:00
What conclusion can you draw from this experiment about the components of the black ink?
Answers: 3

You know the right answer?
What is the MOLAR heat of combustion of methane(CH₄) if 64.00g of methane are burned to heat 75.0 ml...
Questions



Mathematics, 03.06.2020 02:58

Mathematics, 03.06.2020 02:58




English, 03.06.2020 02:58

Mathematics, 03.06.2020 02:58




Mathematics, 03.06.2020 02:58


History, 03.06.2020 02:58

Mathematics, 03.06.2020 02:58


Biology, 03.06.2020 02:58