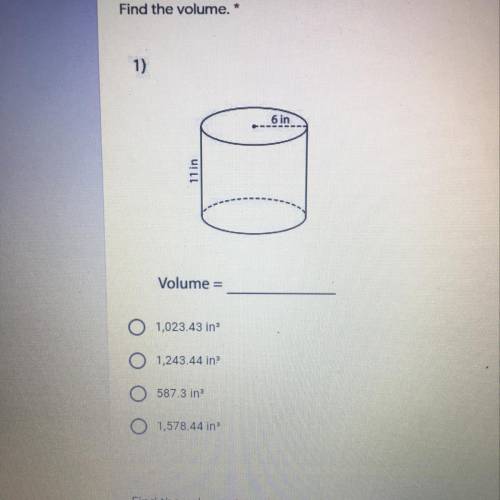
Find the volume. *
1)
6 in
11 in
Volume =
...

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1

Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3

Chemistry, 22.06.2019 14:20
Which of the following are sources of revenue for media companies? a. direct sales to producers b.advertising and subscriptions c. online purchase d. capital investments
Answers: 1

Chemistry, 22.06.2019 19:00
A4.86 g piece of metal was placed in a graduated cylinder containing 15.5 ml of water. the water level rose to 17.3 ml. what is the density of the metal. i need the steps of how to solve it to so i can use a formula to work out other problems.
Answers: 1
You know the right answer?
Questions


Arts, 12.02.2021 19:30


Mathematics, 12.02.2021 19:30


Biology, 12.02.2021 19:30

Mathematics, 12.02.2021 19:30

Physics, 12.02.2021 19:30


History, 12.02.2021 19:30



Social Studies, 12.02.2021 19:30


Mathematics, 12.02.2021 19:30

Mathematics, 12.02.2021 19:30


Chemistry, 12.02.2021 19:30