
Chemistry, 07.05.2020 02:00 linnybear300
Enter your answer in the provided box. Lead(II) nitrate is added slowly to a solution that is 0.0400 M in Cl− ions. Calculate the concentration of Pb2+ ions (in mol / L) required to initiate the precipitation of PbCl2.
(Ksp for PbCl2 is 2.40 × 10−4.)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:30
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 06:30
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 14:30
How can carbon move from "land" to bodies of water? describe the way human impact has lead to increased levels of co2 in the atmosphere.
Answers: 2

Chemistry, 22.06.2019 16:30
At 20°c, a sample of h2o liquid and a sample of co2 gas each have the same average kinetic energy. why is one a liquid and the other a gas at this temperature?
Answers: 1
You know the right answer?
Enter your answer in the provided box. Lead(II) nitrate is added slowly to a solution that is 0.0400...
Questions




Mathematics, 21.02.2020 20:42


Computers and Technology, 21.02.2020 20:43

History, 21.02.2020 20:43


Mathematics, 21.02.2020 20:43


Mathematics, 21.02.2020 20:43



Mathematics, 21.02.2020 20:43


Mathematics, 21.02.2020 20:43



Business, 21.02.2020 20:43

![[Pb^{2+}] } = 0.15 mol/L](/tpl/images/0651/0478/c6ffa.png)
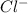 is
is 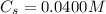

![K_{sp} = [Pb^{2+}][Cl^{-}]^2](/tpl/images/0651/0478/939e9.png)
![2.4 0 * 10^{-4} = 0.0400^2 * [Pb^{2+}]](/tpl/images/0651/0478/b2e7f.png)
![[Pb^{2+}] } = \frac{2.4 0 * 10^{-4} }{0.0400^2 }](/tpl/images/0651/0478/f08bf.png)


