
Sodium (Na) reacts with nitrogen gas (N2) to produce sodium nitride. If a student collects 2.5 moles of sodium nitride (NaN3) after the reaction has been completed, determine the number of moles of nitrogen that were initially present.
2Na + 3N2 -> 2NaN3
Your
3.75 mole N2
1.2 mole N2
3.87 mole N2

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 15:00
Which of the following is the correct formula for copper (i) sulfate trihydrate? cuso4 · 3h2o cuso4(h2o)3 cu2so4(h2o)3 cu2so4 · 3h2o
Answers: 1

Chemistry, 23.06.2019 00:00
What is the approximate mass of 25 cm3 of silver, if the density is 10.5 g/cm3? a. 0.42 g b. 2.4 g c. 42 g d. 260 g
Answers: 1

Chemistry, 23.06.2019 03:00
A0.100-kilogram apple hangs in a tree 1.50 meter above the ground. ignore frictional effects, the total mechanical energy of the apples is
Answers: 1

Chemistry, 23.06.2019 04:00
Why must humans find substitutes for many minerals found on earth? (a) form at an extremely slow rate (b) controlled by other countries (c) too deep in the earth to collect
Answers: 1
You know the right answer?
Sodium (Na) reacts with nitrogen gas (N2) to produce sodium nitride. If a student collects 2.5 moles...
Questions


Mathematics, 16.12.2020 14:00


Mathematics, 16.12.2020 14:00

Geography, 16.12.2020 14:00


Computers and Technology, 16.12.2020 14:00



Mathematics, 16.12.2020 14:00


Arts, 16.12.2020 14:00



English, 16.12.2020 14:00



Mathematics, 16.12.2020 14:00


English, 16.12.2020 14:00

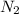
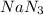 = 2.5 mol
= 2.5 mol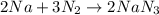
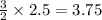 moles of
moles of 

