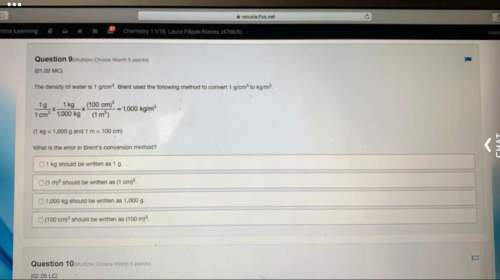
Chemistry, 06.05.2020 20:42 isabellacampos4586
A solution is prepared by dissolving 0.23 mol of lactic acid and 0.27 mol of sodium lactate in water sufficient to yield 1.00 L of solution. The addition of 0.05 mol of HCl to this buffer solution causes the pH to drop slightly. The pH does not decrease drastically because the HCl reacts with the present in the buffer solution. The Ka of lactic acid is 1.36 × 10-3.
A lactate ion
B H2O
C lactic acid
D H3O+
E This is a buffer solution: the pH does not change upon addition of acid or base.

Answers: 2


Another question on Chemistry



Chemistry, 22.06.2019 17:40
How much heat is added if 0.814g of water increase in temperature by 0.351 degree c?
Answers: 3

Chemistry, 23.06.2019 06:20
Examine the false statement. compounds are the smallest unit of an element that occur most commonly in nature. select the rewording of the statement that is true. a: atoms are the smallest unit of an element that commonly occur in nature. b: molecules are the smallest unit of an element or compound that commonly occur in nature. c: molecules are the smallest unit of a compound that occur on the periodic table. d: compounds are the smallest unit of an element that occur on the periodic table
Answers: 1
You know the right answer?
A solution is prepared by dissolving 0.23 mol of lactic acid and 0.27 mol of sodium lactate in water...
Questions

Mathematics, 22.06.2021 05:30

Mathematics, 22.06.2021 05:30


Mathematics, 22.06.2021 05:30



Chemistry, 22.06.2021 05:30




Mathematics, 22.06.2021 05:30

Mathematics, 22.06.2021 05:30

Mathematics, 22.06.2021 05:30



Mathematics, 22.06.2021 05:30

English, 22.06.2021 05:30

Biology, 22.06.2021 05:30

Mathematics, 22.06.2021 05:30