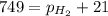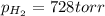Chemistry, 06.05.2020 17:08 22katelynfrankouqqrb
As the magnesium reacts, the hydrogen gas produced is collected by water displacement at 23.0oC. The pressure of the gas in the collection tube is measured to be 749 torr. Given that the equilibrium vapor pressure of water is 21 torr at 23.0oC, calculate the pressure that the H2(g) produced in the reaction would have if it were dry.

Answers: 2


Another question on Chemistry

Chemistry, 20.06.2019 18:04
Scientist wants to react a barium chloride salt with an acid. to speed up the reaction, the scientist crushes the barium chloride salt until it is a fine powder. this speeds up the reaction by a. increasing the ph of the salt. b. increasing the surface area of the salt. c. increasing the pressure of the salt. d. increasing the temperature of the salt. im thinking more a not 100% sure
Answers: 3

Chemistry, 22.06.2019 00:00
Which type of bonding involves the complete transfer of a valence electron from a less electrogrative atom to a more electronegative one
Answers: 1

Chemistry, 22.06.2019 16:00
Is a measure of the resistance to flow. a high liquid has a high resistance to flow and flows slowly. the ancients thought everything in the world was made of 4 we now know that there are 94 naturally occurring and scientists have created another 24 i am certain they will create even more. honey flows slowly because it has a high to flowing. a can be separated by physical means because it contains more than one pure substance and 2 pure substances are not chemically bonded to each other. a cannot be separated by physical means. all matter is made up of all elements are with the same number of protons. if it is just a single or many bonded together, if all of them have the same number of protons, it is an element. in a piece of pure iron metal, all the are joined together, that piece of iron metal is called elemental iron. a single of iron is called elemental iron. a mixture has differences from place to place. we might need a microscope to see them or they might be obvious to the unaided eye. there are surfaces separating it into different phases. a mixture is the same everywhere. it is uniform. there are no surfaces separating it into different phases. if different kinds of atoms (different elements) are bonded together by their electrons, it is called a there are physical means of to isolate the different pure substances in a mixture and there are chemical means of to isolate the different elements in a compound. 1. element 2. compound 3. mixture 4. heterogeneous 5. homogeneous 6. pure substance 7. atoms 8. separation 9. viscosity 10. resistance
Answers: 2

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning explain how a buffer works, using an ethanoic acid/sodium ethanoate system including how the system resists changes in ph upon addition of a small amount of base and upon addition of a small amount of acid respectively. include the following calculations in your i. calculate the ph of a solution made by mixing 25cm3 0.1m ch3cooh and 40cm3 0.1m ch3coo-na+. [ka = 1.74 x 10-5 m] ii. calculate the ph following the addition of a 10cm3 portion of 0.08 m naoh to 500cm3 of this buffer solution. iii. calculate the ph following the addition of a 10cm3 portion of 0.08 m hcl to 200cm3 of the original buffer solution.
Answers: 1
You know the right answer?
As the magnesium reacts, the hydrogen gas produced is collected by water displacement at 23.0oC. The...
Questions

Mathematics, 20.06.2020 20:57

Mathematics, 20.06.2020 20:57

Biology, 20.06.2020 20:57



Mathematics, 20.06.2020 20:57




Mathematics, 20.06.2020 20:57


History, 20.06.2020 20:57




Mathematics, 20.06.2020 20:57




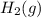 produced in the reaction would have if it were dry will be 728 torr
produced in the reaction would have if it were dry will be 728 torr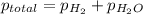
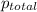 =total pressure of gas = 749 torr
=total pressure of gas = 749 torr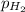 = partial pressure of hydrogen = ?
= partial pressure of hydrogen = ?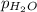 = partial pressure of water = 21 torr
= partial pressure of water = 21 torr