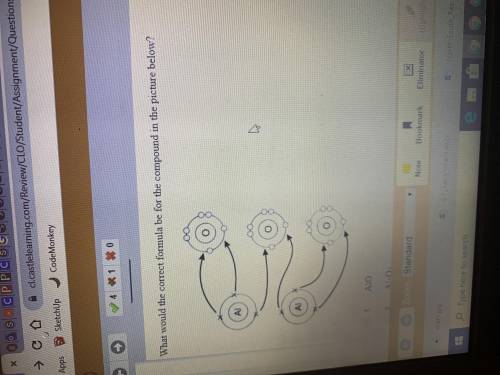
What is the correct formula for the compound in the picture
...

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1


Chemistry, 22.06.2019 23:30
The density of benzene at 15 °c is 0.8787 g/ml. calculate the mass of 0.1500 l of benzene at this temperature. enter your answer in terms of grams
Answers: 2
You know the right answer?
Questions

English, 05.05.2020 13:49

Mathematics, 05.05.2020 13:49

Computers and Technology, 05.05.2020 13:49



Mathematics, 05.05.2020 13:49

History, 05.05.2020 13:49


English, 05.05.2020 13:49



Mathematics, 05.05.2020 13:49


Mathematics, 05.05.2020 13:49


Mathematics, 05.05.2020 13:49

Biology, 05.05.2020 13:49


Health, 05.05.2020 13:49