120 grams of HF are needed to react with 3.0 moles of Sn
Explanation:
The balanced chemical equation is :
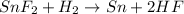
According to stoichiometry:
1 mole of 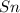 reacts with 2 moles of HF
reacts with 2 moles of HF
Thus 3.0 moles of Sn will react = 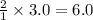 moles of HF
moles of HF
Mass of 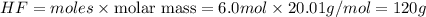
Thus 120 grams of HF are needed to react with 3.0 moles of Sn



























