Chemistry, 05.05.2020 20:44 budjasdatazaki3913
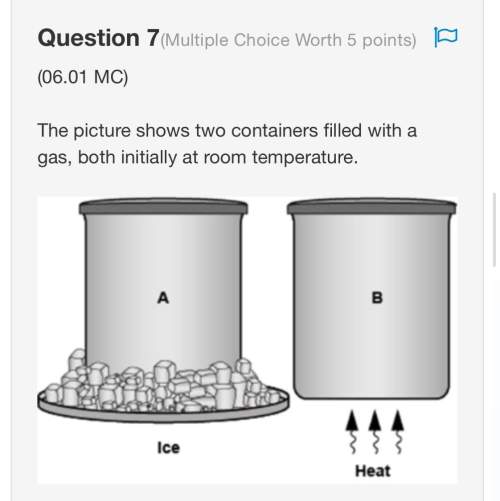
The graph shows the volume of a gaseous product formed during two trials of a reaction. A different concentration of reactant was used during each trial, whereas the other factors were kept constant.
A graph is shown with two graph lines sloping upwards. The title on the x axis is Time and the title on the y axis is Amount of Product. The graph line with a steeper slope is labeled Trial 2 and the other is labeled Trial 1.
Which of the following statements explains which trial has a lower concentration of the reactant?
Trial 1, because the average rate of the reaction is lower.
Trial 1, because this reaction lasted for a longer duration than Trial 2.
Trial 2, because this reaction was initially fast and later slowed down.
Trial 2, because the volume of product formed per unit time was higher.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 02:20
6. what does the symbol ah stand for? o one calorie given off by a reaction the specific heat of a substance the heat capacity of a substance the heat of reaction for a chemical reaction
Answers: 1

Chemistry, 22.06.2019 04:00
Electric charge is what ? a. kinetic energy b. radiation c. discovery d. electricity
Answers: 1

Chemistry, 22.06.2019 14:20
Which of the following are sources of revenue for media companies? a. direct sales to producers b.advertising and subscriptions c. online purchase d. capital investments
Answers: 1

Chemistry, 22.06.2019 21:00
One similarity and one difference between an element and a mixture of elements
Answers: 1
You know the right answer?
The graph shows the volume of a gaseous product formed during two trials of a reaction. A different...
Questions


Mathematics, 24.10.2021 21:40

Mathematics, 24.10.2021 21:40

Chemistry, 24.10.2021 21:40

Mathematics, 24.10.2021 21:40

Mathematics, 24.10.2021 21:40

Social Studies, 24.10.2021 21:40

Computers and Technology, 24.10.2021 21:40

World Languages, 24.10.2021 21:40




Mathematics, 24.10.2021 21:40

World Languages, 24.10.2021 21:40

Mathematics, 24.10.2021 21:40

Chemistry, 24.10.2021 21:40


Social Studies, 24.10.2021 21:40