An oxide of aluminum contains 0.545 g of Al and 0.485 g of O. Find the empirical formula
for t...

Chemistry, 05.05.2020 09:45 carolyntowerskemp
An oxide of aluminum contains 0.545 g of Al and 0.485 g of O. Find the empirical formula
for the oxide.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:00
In any energy conversion, some of the energy is lost to the environment as question 5 options: electrical energy potential energy sound energy thermal energy
Answers: 1

Chemistry, 22.06.2019 16:40
The diagram below shows the movement of particles. what does this piece of evidence best support? the collision theory the maxwell-boltzmann distribution the effect of pressure on reaction rates the effect of temperature on reaction rates
Answers: 3


Chemistry, 22.06.2019 22:30
Which process describes vaporization that takes place below the surface of a liquid? condensation melting boiling evaporation
Answers: 1
You know the right answer?
Questions



Mathematics, 10.02.2021 06:20

English, 10.02.2021 06:20

Business, 10.02.2021 06:20





Advanced Placement (AP), 10.02.2021 06:20

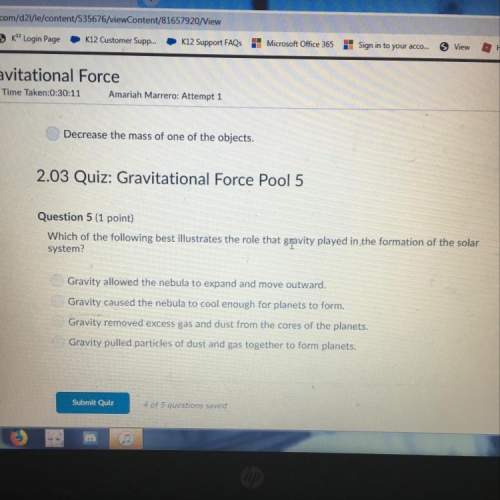
Chemistry, 10.02.2021 06:20

English, 10.02.2021 06:20


Mathematics, 10.02.2021 06:20

Mathematics, 10.02.2021 06:20

English, 10.02.2021 06:20

Mathematics, 10.02.2021 06:20



Biology, 10.02.2021 06:20