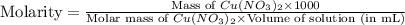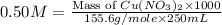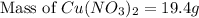Which calculation below represents the mass of
copper(II) nitrite (MM = 155.6 g/mol) should be...

Chemistry, 05.05.2020 04:15 barnettboy76
Which calculation below represents the mass of
copper(II) nitrite (MM = 155.6 g/mol) should be
weighed out to produce 250 ml of a 0.50 M solution?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:20
Which of the following statements is false regarding aromaticity? a. the compound must be cyclic b. the compound must be fully conjugated c. the compound must be planar d.the number of electrons in the pi system must satisfy the hückel 4n+2 rule e. the compound must have a neutral charge
Answers: 2

Chemistry, 22.06.2019 09:50
Achemist has dissolved a certain substance in water. the chemist knows that more of the substance could be dissolved into the water before it stops dissolving. therefore
Answers: 2

Chemistry, 22.06.2019 11:30
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2

Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2
You know the right answer?
Questions

Mathematics, 07.12.2021 01:40


Mathematics, 07.12.2021 01:40


Mathematics, 07.12.2021 01:40

History, 07.12.2021 01:40



History, 07.12.2021 01:40





History, 07.12.2021 01:40

Mathematics, 07.12.2021 01:40


Arts, 07.12.2021 01:40


Mathematics, 07.12.2021 01:40

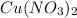 in the solution is, 19.4 grams.
in the solution is, 19.4 grams.