Chemistry, 05.05.2020 07:10 ghanim1963
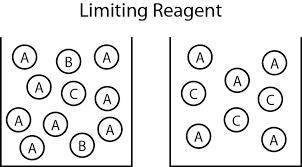
An aqueous solution containing 9.82 g9.82 g of lead(II) nitrate is added to an aqueous solution containing 5.76 g5.76 g of potassium chloride. Enter the balanced chemical equation for this reaction. Be sure to include all physical states. balanced chemical equation: Pb(NO3)2(aq)+2KCl(aq)⟶PbCl2(s)+2KNO 3(aq)Pb(NO3)2(aq)+2KCl(aq)⟶PbCl2(s) +2KNO3(aq) What is the limiting reactant? potassium chloride lead(II) nitrate The percent yield for the reaction is 87.5%87.5% . How many grams of the precipitate are formed? precipitate formed: gg How many grams of the excess reactant remain?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:30
Determine the reaction and balance the following equations urgent due in the morning
Answers: 2


Chemistry, 22.06.2019 22:30
What relationship exists between an enzyme and a catalyst?
Answers: 1

Chemistry, 22.06.2019 23:00
What is the energy in joules of a mole of photons associated with visible light of wavelength 486 nm?
Answers: 3
You know the right answer?
An aqueous solution containing 9.82 g9.82 g of lead(II) nitrate is added to an aqueous solution cont...
Questions





Health, 25.06.2019 17:30

Mathematics, 25.06.2019 17:30

English, 25.06.2019 17:30

Biology, 25.06.2019 17:30

Mathematics, 25.06.2019 17:30









Computers and Technology, 25.06.2019 17:30


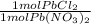 *
*  * 87.5/100 = 7.20 g PbCl₂
* 87.5/100 = 7.20 g PbCl₂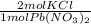 *
* 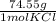 = 3.86 g KCl
= 3.86 g KCl