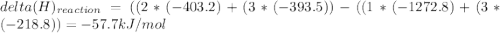Chemistry, 05.05.2020 08:30 jackiediaz
Use the given heats of formation to calculate the enthalpy change for this reaction. B2O3(g) + 3COCl2(g) →2BCl3(g) + 3CO2(g) ΔHof of B2O3(g) is –1272.8 kJ∙mol–1 ΔHof of BCl3(g) is –403.8 kJ∙mol–1 ΔHof of COCl2(g) is –218.8 kJ∙mol–1 ΔHof of CO2(g) is –393.5 kJ∙mol–1 Question 8 options: 1) 649.3 kJ·mol–1 2) 354.9 kJ·mol–1 3) –58.9 kJ·mol–1 4) –3917.3 kJ·mol–1

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Which type of bonding involves the complete transfer of a valence electron from a less electrogrative atom to a more electronegative one
Answers: 1

Chemistry, 22.06.2019 03:00
About 70 percent of the earth's surface is water-covered, and about 96.5 percent of all earth's water is salt water. identify the watery feature on earth that is made of freshwater rather than salt water. a) bay b) glacier c) ocean d) sea it is not incomplete this is the true question
Answers: 1

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 07:30
What three things determine the shape and size of a puddle when water is poured out onto a surface
Answers: 2
You know the right answer?
Use the given heats of formation to calculate the enthalpy change for this reaction. B2O3(g) + 3COCl...
Questions


Physics, 18.12.2021 15:20

Computers and Technology, 18.12.2021 15:20

Mathematics, 18.12.2021 15:20

Mathematics, 18.12.2021 15:20

Mathematics, 18.12.2021 15:20


Mathematics, 18.12.2021 15:20

History, 18.12.2021 15:20








Social Studies, 18.12.2021 15:30

Social Studies, 18.12.2021 15:30


Biology, 18.12.2021 15:40