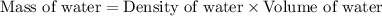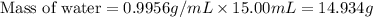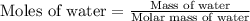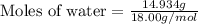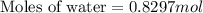that has a density of 0.9956 g/ ml.

Chemistry, 05.05.2020 12:41 kyasnead8189
How many moles of water are present in 15.00 ml of water
that has a density of 0.9956 g/ ml.
MW for Hydrogen 1.00 g/ 1 mole; MW for Oxygen 16.00 g/ 1 mole

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 14:40
What type of solution is formed if 10 g of kclo3 are dissolved in 100g of water at 30
Answers: 2

Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2

Chemistry, 22.06.2019 03:20
What is the ima of the 1 st class lever in the graphic given? 2 3 0.5
Answers: 1

Chemistry, 23.06.2019 11:20
Which of the following is a pure substance? airbloodcopperwood
Answers: 2
You know the right answer?
How many moles of water are present in 15.00 ml of water
that has a density of 0.9956 g/ ml.
that has a density of 0.9956 g/ ml.
Questions

Social Studies, 22.09.2019 09:00



English, 22.09.2019 09:00



History, 22.09.2019 09:00

History, 22.09.2019 09:00



Mathematics, 22.09.2019 09:00

Mathematics, 22.09.2019 09:00

Social Studies, 22.09.2019 09:00

Biology, 22.09.2019 09:00

Mathematics, 22.09.2019 09:00

Arts, 22.09.2019 09:00


Biology, 22.09.2019 09:00


History, 22.09.2019 09:00