Chemistry, 05.05.2020 14:58 mccay5016987
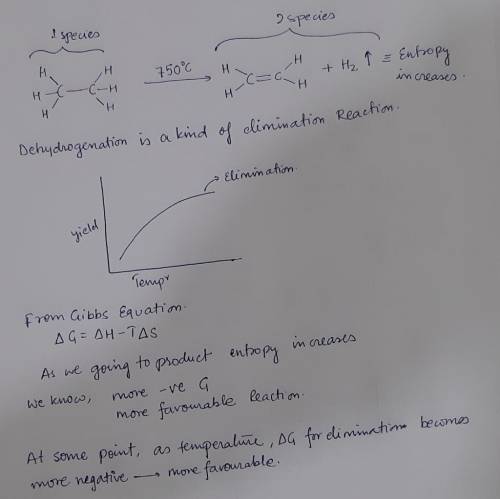
6. At high temperatures, alkanes can undergo dehydrogentation to produce alkenes. This reaction is used industrially to prepare ethylene while simultaneously serving as a source of hydrogen gas. Explain why dehydrogenation only works at high temperatures using approximately 20 words or less. (5 poin

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:40
Determine the energy released per kilogram of fuel used. given mev per reaction, calculate energy in joules per kilogram of reactants. consider 1 mole of tritium plus 1 mole of deuterium to be a mole of “reactions” (total molar mass = 5 grams).
Answers: 1

Chemistry, 22.06.2019 09:40
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1

Chemistry, 22.06.2019 11:30
If we compare and contrast electromagnetic waves with sound waves, all but one statement is true. that is a) sound waves require a medium to travel while electromagnetic waves do not. b) electromagnetic waves can travel through the vacuum of space while sound waves cannot. c) electromagnetic waves must have a medium in which to travel, but sound waves can travel anywhere. eliminate d) sound waves must bounce off of matter in order to travel while electromagnetic waves do not require matter to be present.
Answers: 3

Chemistry, 22.06.2019 12:00
Which of the following units is not an official si unit? mole liter kilogram ampere
Answers: 1
You know the right answer?
6. At high temperatures, alkanes can undergo dehydrogentation to produce alkenes. This reaction is u...
Questions



Social Studies, 31.08.2019 04:30


Mathematics, 31.08.2019 04:30



Mathematics, 31.08.2019 04:30


History, 31.08.2019 04:30



Social Studies, 31.08.2019 04:30

Health, 31.08.2019 04:30

Computers and Technology, 31.08.2019 04:30




Geography, 31.08.2019 04:30