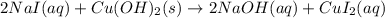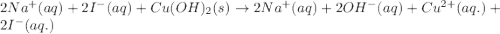Chemistry, 05.05.2020 16:26 rondonalba
Part 1: Write the complete balanced MOLECULAR equation (including all states of matter) for the precipitation reaction that occurs between aqueous copper(II) chloride and aqueous sodium hydroxide.
Part 2: Write the NET IONIC EQUATION for the REVERSE reaction in Part 1 (include all states of matter).
Part 3: Write the equilibrium expression for the net ionic equation written in Part 2.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:50
2. you__turn left on a red light if you are in the left-most lane of a one-way street, you're turning into the left-most lane of a one-way street, and no nearby sign prohibits the turn.
Answers: 2


Chemistry, 22.06.2019 14:30
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1

Chemistry, 22.06.2019 23:30
The ammonia molecule in the diagram has the observed bond orientation because
Answers: 1
You know the right answer?
Part 1: Write the complete balanced MOLECULAR equation (including all states of matter) for the prec...
Questions

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Biology, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

English, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

Mathematics, 16.09.2020 16:01

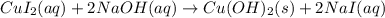
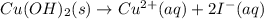
![K_{eq}=[Cu^{2+}][I^-]^2](/tpl/images/0640/1859/a2d62.png)