
Chemistry, 05.05.2020 18:18 mshepherdmiller
An aqueous solution has [C6H5COOH] = 0.110 M and [Ca(C6H5COO)2] = 0.200 M. Ka = 6.3 × 10-5 for C6H5COOH. The solution volume is 5.00 L. What is the pH of the solution after 10.00 mL of 5.00 M NaOH is added? Group of answer choices 4.81 4.86 4.75 4.70 4.65

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
How many atoms of mg are present in 97.22 grams of mg? 6.022 × 1023 2.408 × 1024 4.818 × 1024 5.855 × 1025
Answers: 2

Chemistry, 22.06.2019 12:00
the mississippians were considered to be horticulturalists, which means they were
Answers: 1

Chemistry, 22.06.2019 13:30
What are the chemical names of these compounds? ke: mg3n2: reset next
Answers: 1

Chemistry, 22.06.2019 20:30
Water undergoes a large change in density at 0 ∘ c as it freezes to form ice. calculate the percent change in density that occurs when liquid water freezes to ice at 0 ∘ c given that
Answers: 2
You know the right answer?
An aqueous solution has [C6H5COOH] = 0.110 M and [Ca(C6H5COO)2] = 0.200 M. Ka = 6.3 × 10-5 for C6H5C...
Questions

Mathematics, 13.03.2021 02:30


Mathematics, 13.03.2021 02:30

History, 13.03.2021 02:30

Mathematics, 13.03.2021 02:30


Spanish, 13.03.2021 02:30


Mathematics, 13.03.2021 02:30

Mathematics, 13.03.2021 02:30

Chemistry, 13.03.2021 02:30

Chemistry, 13.03.2021 02:30



Mathematics, 13.03.2021 02:30


Mathematics, 13.03.2021 02:30


Mathematics, 13.03.2021 02:30

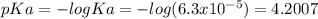
![pH=pKa+log\frac{C6H5COO]}{[C6H5COOH]} =4.2007+log\frac{0.4}{0.11} =4.7614](/tpl/images/0641/1671/7b407.png)
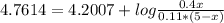
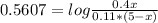
![An aqueous solution has [C6H5COOH] = 0.110 M and [Ca(C6H5COO)2] = 0.200 M. Ka = 6.3 × 10-5 for C6H5C](/tpl/images/0641/1671/b03ac.jpg)



