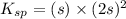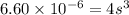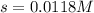Chemistry, 05.05.2020 23:02 sophiateaches053
The ksp of pbbr2 is 6.60×10−6. What is the molar solubility of pbbr2 in pure water?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Identify the missing numbers below to show the result of multiplying the numbers (1.6 × 10-19)(5.0 × 106) = c × 10d
Answers: 1


Chemistry, 22.06.2019 22:30
Which of the following molecules is polar? c3h7oh c2h5cooh
Answers: 1

Chemistry, 23.06.2019 04:00
Calculate the mass of 0.750 mol of the following substance. na3po4. , i'm not quite sure on how to set up the problem to solve! : (
Answers: 1
You know the right answer?
The ksp of pbbr2 is 6.60×10−6. What is the molar solubility of pbbr2 in pure water?...
Questions

Biology, 10.07.2019 16:30


Biology, 10.07.2019 16:30




History, 10.07.2019 16:30


Business, 10.07.2019 16:30

History, 10.07.2019 16:30


Social Studies, 10.07.2019 16:30


Social Studies, 10.07.2019 16:30

Social Studies, 10.07.2019 16:30

Business, 10.07.2019 16:30



Biology, 10.07.2019 16:30

Business, 10.07.2019 16:30

 in pure water is, 0.0118 M
in pure water is, 0.0118 M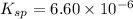
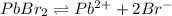
![K_{sp}=[Pb^{2+}][Br^{-}]^2](/tpl/images/0643/5049/61d34.png)