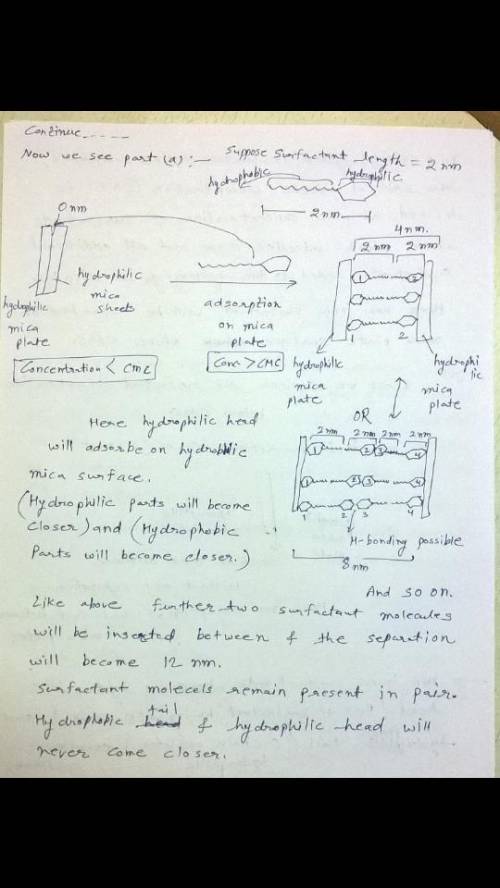
A researcher measures the equilibrium separation between two flat hydrophilic mica plates in a solution of non-ionic surfactant. When the surfactant concentration is below CMC the surfaces touch at 0 nm distance without any separation (i. e., there is no surfactant adsorption and repulsion)
a) The researcher increases the surfactant concentration much above CMC and finds out that the system has equilibrium separations at 0 nm, 4 nm, 8 nm, 12 nm and some at larger distances. Draw schematics and explain the reason for this effect.
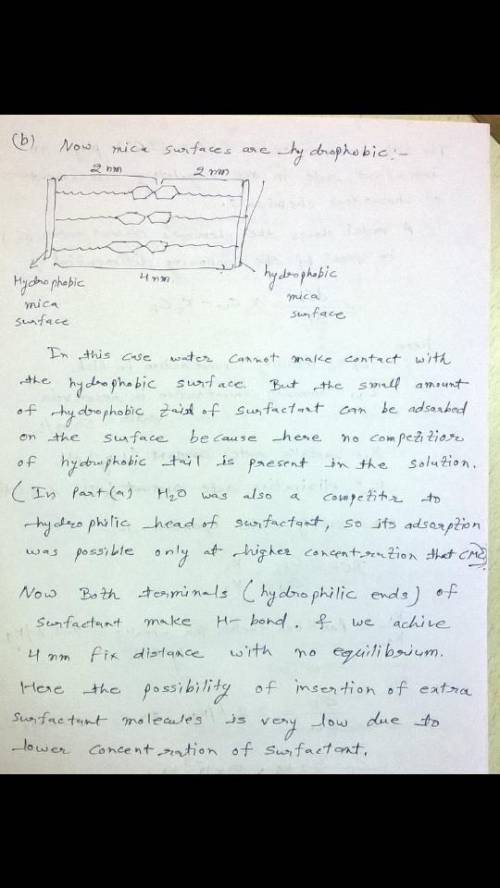
b) The researcher hydrophobizes the mica surfaces and again dips them in the first solution with surfactant concentration below CMC. When these surfaces are brought in contact, they remain separated at a distance of 4 nm (no equilibria at larger separations). Draw a schematic and explain the reason for this observation with hydrophobic surfaces.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 1

Chemistry, 22.06.2019 16:10
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1

You know the right answer?
A researcher measures the equilibrium separation between two flat hydrophilic mica plates in a solut...
Questions

English, 02.02.2021 22:50





Spanish, 02.02.2021 22:50


Mathematics, 02.02.2021 22:50


English, 02.02.2021 22:50

Mathematics, 02.02.2021 22:50


Mathematics, 02.02.2021 22:50

Mathematics, 02.02.2021 22:50

History, 02.02.2021 22:50

Chemistry, 02.02.2021 22:50

Mathematics, 02.02.2021 22:50


Mathematics, 02.02.2021 22:50

Social Studies, 02.02.2021 22:50