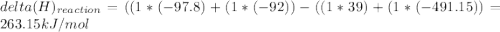Chemistry, 06.05.2020 06:36 jetblackcap
Hydroxyl radicals react with and eliminate many atmospheric pollutants. However, the hydroxyl radical does not clean up everything. For example, chlorofluorocarbons - which destroy stratospheric ozone - are not attacked by the hydroxyl radical. Consider the hypothetical reaction by which the hydroxyl radical might react with a chlorofluorocarbon: OH(g)+CF2Cl2(g)-->HOF(g)+CFCl2(g )Use bond energies to explain why this reaction is improbable. Calculate \Delta Hrxn of this reaction.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Select all that apply. a beta particle: is electromagnetic energy is an electron has zero charge is emitted from the nucleus has a +2 charge has a -1 charge
Answers: 1

Chemistry, 22.06.2019 14:00
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1

Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1

Chemistry, 22.06.2019 19:40
Scientists have developed an explanation of a phenomenon from several verified hypotheses. the explanation has been confirmed through numerous experimental tests.which option best describes this explanation? a. scientific lawb. research questionc. hypothesisd. scientific theory
Answers: 3
You know the right answer?
Hydroxyl radicals react with and eliminate many atmospheric pollutants. However, the hydroxyl radica...
Questions

Mathematics, 16.12.2021 02:40


Mathematics, 16.12.2021 02:40

History, 16.12.2021 02:40








Mathematics, 16.12.2021 02:40





Mathematics, 16.12.2021 02:40