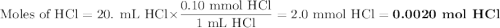Chemistry, 06.05.2020 07:17 gracelong4326
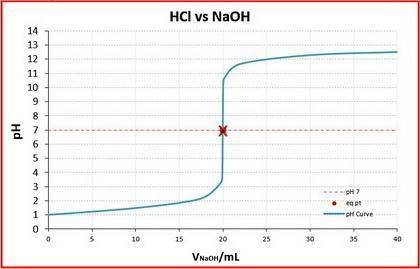
A student was given the task of titrating a 20.mL sample of 0.10MHCl(aq) with 0.10MNaOH(aq) . The HCl(aq) was placed in an Erlenmeyer flask. An equation for the reaction that occurs during the titration is given above.
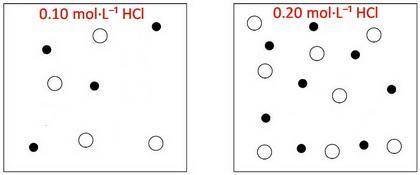
The box below to the left represents ions in a certain volume of 0.10MHCl(aq) . In the box below to the right, draw a representation of ions in the same volume of 0.20MHCl(aq)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:00
The alkali metals (group 1) consist of lithium (3), sodium (11), potassium (19), rubidium (37), cesium (55), and francium (87). they are soft, metallic solids with low densities and low melting points. based on the data shown in figure 1, how many valence electrons do alkali metals share?
Answers: 3

Chemistry, 22.06.2019 06:00
Calculate the mass of silver needed to react with chlorine to produce 126g if silver chloride?
Answers: 3

Chemistry, 22.06.2019 13:00
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3

Chemistry, 22.06.2019 13:10
Select the correct answer a modure consists of glucose and water. what is the percent composition of glucose in the mixture if it contains 1.3 moles of glucose (cho total mass of the mature is 276 grams? ) and the a 1775
Answers: 1
You know the right answer?
A student was given the task of titrating a 20.mL sample of 0.10MHCl(aq) with 0.10MNaOH(aq) . The HC...
Questions


History, 18.07.2019 15:30




Chemistry, 18.07.2019 15:30




Spanish, 18.07.2019 15:30










Advanced Placement (AP), 18.07.2019 15:30