
Chemistry, 25.04.2020 23:51 gabrielolivas59
C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O (l)
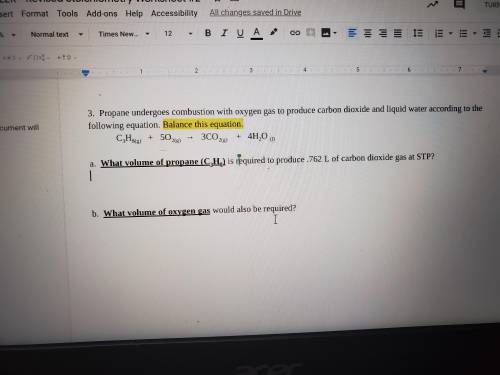
a. What volume of propane (C3H8) is required to produce .762 L of carbon dioxide gas at STP?
b. What volume of oxygen gas would also be required?

Answers: 1


Another question on Chemistry



Chemistry, 23.06.2019 03:30
If 2 molecules of one reactant combine with 3 molecules of another to produce 5 molecules of a product, then what is the representation of the reaction?
Answers: 1

You know the right answer?
C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O (l)
a. What volume of propane (C3H8) is required to pr...
a. What volume of propane (C3H8) is required to pr...
Questions


English, 22.04.2021 17:40

Mathematics, 22.04.2021 17:40



Biology, 22.04.2021 17:40

English, 22.04.2021 17:40



Mathematics, 22.04.2021 17:40



Mathematics, 22.04.2021 17:40






Mathematics, 22.04.2021 17:40

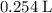 (at STP.)
(at STP.)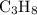 and
and 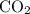 act like ideal gases.
act like ideal gases.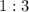 . As a result, for every
. As a result, for every 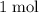 of
of 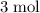 of
of 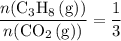 .
.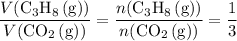 .
.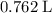 of
of 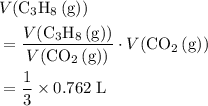 .
.

