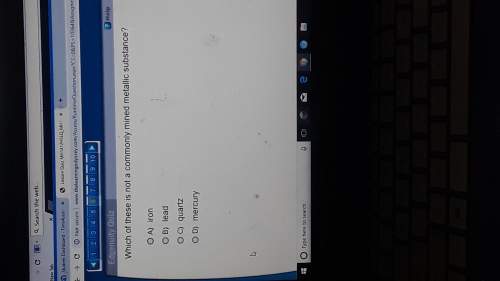
Chemistry, 24.04.2020 18:31 kylierice1
Determined the order of the reaction with respect to IO3- to be 1 and 1 respectively. The actual order of the reaction with respect to IO3- is 1. This along with your value for the order of the reaction with respect to S2O52- gives a rate law for the reaction of rate=k[IO3-]1[S2O52-]1.3

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 05:30
What happens to the atomic radius when an elctron is lost
Answers: 1

Chemistry, 22.06.2019 14:30
Amixture that has two or more substances that are spread out evenly is called a. compound b. heterogeneous c. substance d. homogeneous
Answers: 1

Chemistry, 22.06.2019 19:00
What is the compound name for the formula [ru(en)2cl2]2+ and [co(en)cl2br]-
Answers: 1
You know the right answer?
Determined the order of the reaction with respect to IO3- to be 1 and 1 respectively. The actual ord...
Questions

Biology, 14.07.2019 20:30

History, 14.07.2019 20:30


Biology, 14.07.2019 20:30

Mathematics, 14.07.2019 20:30

Business, 14.07.2019 20:30


History, 14.07.2019 20:30

Mathematics, 14.07.2019 20:30





English, 14.07.2019 20:30


Social Studies, 14.07.2019 20:30


Mathematics, 14.07.2019 20:30

Physics, 14.07.2019 20:30