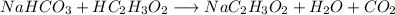Chemistry, 24.04.2020 01:54 brionesoswaldo09
You want to help your little brother make an exploding volcano for his science class. The lava will be made from reacting baking soda (NaHCO3) with vinegar (HC2H3O2). After building the volcano, you know that you want to create about 100.0g of lava (or sodium acetate, NaC2H3O2). Too little lava, and the volcano won’t overflow. Too much lava would be a giant mess! Using stoichiometry and the equation below, calculate the exact amount of baking soda needed to make 100.0g of lava. Assume you have excess vinegar. NaHCO3 + HC2H3O2 → NaC2H3O2 + H2O + CO2

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 13:20
Determine which intermolecular forces are the dominant (strongest) forces for a pure sample of each of the following molecules by placing the molecules into the correct bins. drag the appropriate molecular formula to their respective bins.
Answers: 3


Chemistry, 22.06.2019 00:00
Explain which group an element with the electron configuration 1s2 2s2 2p6 3s2 3p6 3d1 4s2 belongs to.
Answers: 3

Chemistry, 22.06.2019 11:00
What is the molar mass of a gas that has density of 2.054 g/l
Answers: 2
You know the right answer?
You want to help your little brother make an exploding volcano for his science class. The lava will...
Questions

Mathematics, 15.11.2021 01:50


Social Studies, 15.11.2021 01:50

Geography, 15.11.2021 01:50

Mathematics, 15.11.2021 01:50



History, 15.11.2021 01:50

Mathematics, 15.11.2021 01:50

Mathematics, 15.11.2021 01:50

Social Studies, 15.11.2021 01:50

English, 15.11.2021 01:50

Mathematics, 15.11.2021 01:50

Mathematics, 15.11.2021 01:50




Mathematics, 15.11.2021 02:00