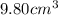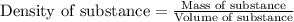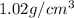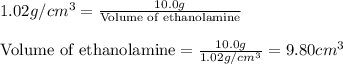Chemistry, 23.04.2020 02:10 abigailweeks10
A chemistry student needs 10.0g of ethanolamine for an experiment. By consulting the CRC Handbook of Chemistry and Physics, the student discovers that the density of ethanolamine is ·1.02gcm−3. Calculate the volume of ethanolamine the student should pour out.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Select the correct answer. given: 2libr + ba → babr2 + 2li in this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced? a. 1.18 mol b. 2.37 mol c. 4.73 mol d. 16.4 mol e. 32.9 mol
Answers: 2

Chemistry, 22.06.2019 04:40
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 07:00
At 450 mm hg a gas has a volume of 760 l, what is its volume at standard pressure
Answers: 2

Chemistry, 23.06.2019 00:00
What is the empirical formula of a compound that is 50.7% antimony and 49.3% selenium ?
Answers: 2
You know the right answer?
A chemistry student needs 10.0g of ethanolamine for an experiment. By consulting the CRC Handbook of...
Questions



Arts, 01.08.2019 22:00






History, 01.08.2019 22:00





Mathematics, 01.08.2019 22:00


English, 01.08.2019 22:00

History, 01.08.2019 22:00