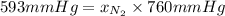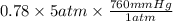Chemistry, 22.04.2020 23:16 juanitarodrigue
The partial pressure of N2 in the air is 593 mm Hg at 1 atm. What is the partial pressure of N2 in a bubble of air a scuba diver breathes when he is 132 ft below the surface of the water where the pressure is 5.00 atm?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:00
What pressure will be exerted by 0.675 moles of a gas at 25*c if it is in a 0.750-l container?
Answers: 1

Chemistry, 22.06.2019 06:30
Particle model to predict what will happen if a sharp object creates a hole in the soccer ball
Answers: 2


Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
The partial pressure of N2 in the air is 593 mm Hg at 1 atm. What is the partial pressure of N2 in a...
Questions


Physics, 27.04.2020 02:23


Chemistry, 27.04.2020 02:23


Chemistry, 27.04.2020 02:23


Mathematics, 27.04.2020 02:23

Biology, 27.04.2020 02:23

Chemistry, 27.04.2020 02:23


English, 27.04.2020 02:23




Mathematics, 27.04.2020 02:23



Mathematics, 27.04.2020 02:23

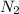 at a depth of 132 ft below sea level is 2964 mm Hg.
at a depth of 132 ft below sea level is 2964 mm Hg.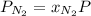
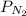 = partial pressure of
= partial pressure of 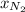 = mole fraction of
= mole fraction of