Chemistry, 22.04.2020 21:55 felipeee4609
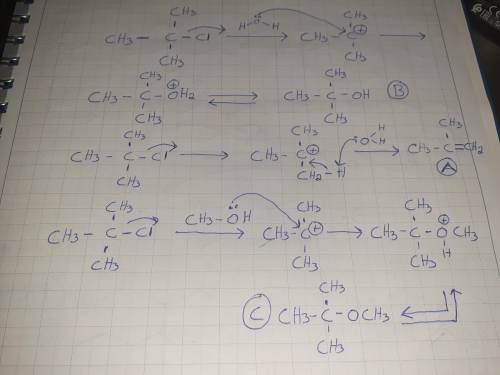
Treating (CH3)3C-Cl with a mixture of H2O and CH3OH at room temperature would yield: A) CH2=C(CH3)2 B) (CH3)3COH C) (CH3)3COCH3 D) All of these choices. E) None of these choices.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2


Chemistry, 22.06.2019 12:00
What is the percentage of hydrogen in nitrogen trihydride
Answers: 1

Chemistry, 22.06.2019 20:30
Citric acid has a ph between 1 and 3. it is considered to be aa. weak acidb. weak basec. strong based. strong acid
Answers: 2
You know the right answer?
Treating (CH3)3C-Cl with a mixture of H2O and CH3OH at room temperature would yield: A) CH2=C(CH3)2...
Questions





Social Studies, 25.12.2019 21:31

Mathematics, 25.12.2019 21:31




Chemistry, 25.12.2019 21:31



Chemistry, 25.12.2019 21:31

History, 25.12.2019 21:31

Mathematics, 25.12.2019 21:31



Spanish, 25.12.2019 21:31

Biology, 25.12.2019 21:31