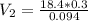Chemistry, 22.04.2020 19:52 blink182lovgabbie
During your reaction, you added 0.3 mL concentrated H2SO4 (18.4 M) as the catalyst. At the end of the reaction, you need to add base to neutralize it. How much volume (in mL) of 10% Na2CO3 to neutralize all the acid present

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
13. calculate the initial concentration (before precipitation) of carbonate ions after the addition of each 0.05 ml of solution b to the 1.00 l beaker of solution a. divide the work among group members and write the answers in the table in model 3. assume the volume change as solution b is added is negligible. 14. notice the initial concentrations of zn2+ - and cu2+ in the table in model 3. a. explain how these were obtained from the data in model 2. b. as solution b is added and precipitates form, do these initial concentrations change? 15. use the data in model 2 to indicate the presence of precipitate (either znco3 or cuco3) after each 0.05 ml addition of solution b in model 3. 16. use the initial concentrations of carbonate ions and zinc ions to calculate the reaction quotient, qsp for the zinc carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3. 17. use the initial concentrations of carbonate ion and copper(ii) ions to calculate the qsp for the copper(ii) carbonate scenarios in model 3. divide the work among group members and write the answers in the table in model 3.
Answers: 3

Chemistry, 22.06.2019 10:30
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2

Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1

Chemistry, 22.06.2019 14:50
Which of the following is most likely true about water in chemical solutions?
Answers: 1
You know the right answer?
During your reaction, you added 0.3 mL concentrated H2SO4 (18.4 M) as the catalyst. At the end of th...
Questions

English, 22.04.2020 12:47


Arts, 22.04.2020 12:47

Geography, 22.04.2020 12:47



Mathematics, 22.04.2020 12:48


Mathematics, 22.04.2020 12:48



History, 22.04.2020 12:48

Mathematics, 22.04.2020 12:48


Biology, 22.04.2020 12:52

Physics, 22.04.2020 12:52

Mathematics, 22.04.2020 12:59

Chemistry, 22.04.2020 12:59


Geography, 22.04.2020 12:59