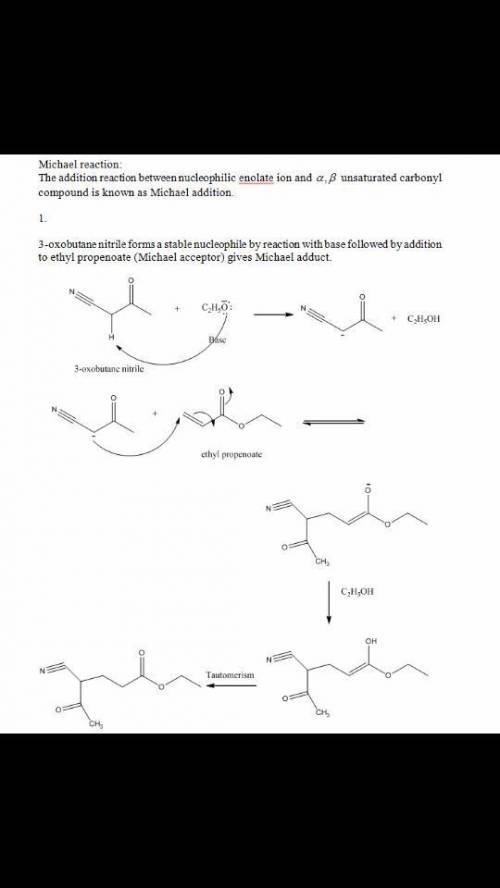
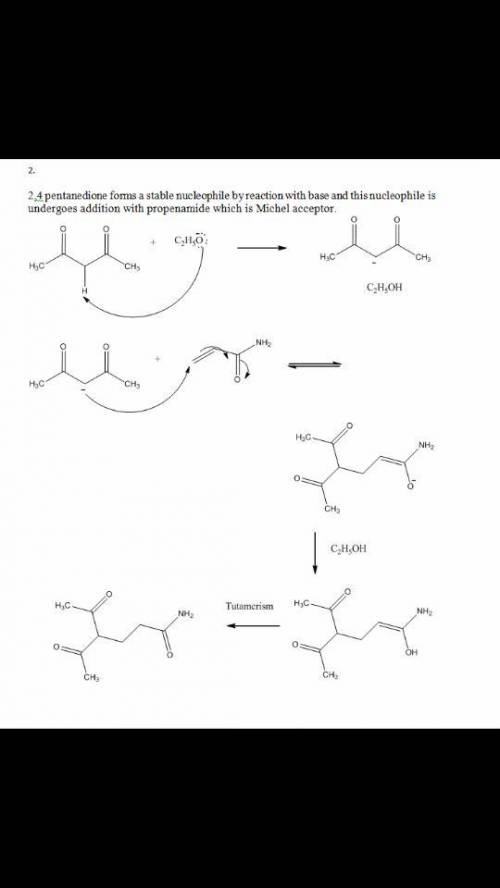
Chemistry, 22.04.2020 04:52 alyssamaize
Please help! The Michael reaction is a conjugate addition reaction between a stable nucleophilic enolate ion (the donor) and an α,β-unsaturated carbonyl compound (the acceptor). 1.) Draw the structure of the product of the Michael reaction between ethyl propenoate and 3-oxobutanenitrile.2.)The Michael reaction is a conjugate addition reaction between a stable nucleophilic enolate ion (the donor) and an α,β-unsaturated carbonyl compound (the acceptor). Draw the structure of the product of the Michael reaction between propenamide and 2,4-pentanedione.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 20:10
What would happen to a volleyball left outside in the winter? o o o o a. it would expand. b. it would lose air. c. it would shrink. d. it would explode.
Answers: 2

Chemistry, 22.06.2019 21:30
If 22.5 of nitrogen at 748 mm hg are compressed to 725 mm hg at constant temperature. what is the new volume?
Answers: 1

Chemistry, 23.06.2019 04:31
Areaction is first order. if the initial reactant concentration is 0.0200 m, and 25.0 days later the concentration is 6.25 x 10-4 m, then its half-life is:
Answers: 1
You know the right answer?
Please help! The Michael reaction is a conjugate addition reaction between a stable nucleophilic eno...
Questions

Biology, 13.09.2019 01:30

Chemistry, 13.09.2019 01:30





Physics, 13.09.2019 01:30

History, 13.09.2019 01:30



Health, 13.09.2019 01:30

Mathematics, 13.09.2019 01:30


Mathematics, 13.09.2019 01:30


Mathematics, 13.09.2019 01:30

Mathematics, 13.09.2019 01:30

English, 13.09.2019 01:30

Social Studies, 13.09.2019 01:30

Biology, 13.09.2019 01:30