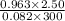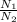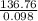You have a 25.0 L cylinder of helium at a pressure of 132 atm and a temperature of 19 [infinity]C. The He is used to fill balloons to a volume of 2.50 L at 732 mm Hg and 27 [infinity]C. How many balloons can be filled with He? Assume that the cylinder can provide He until its internal pressure reaches 1.00 atm (i. e., there are 131 atmospheres of usable He in the cylinder).

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:40
Kc = 0.040 for the system below at 450oc. if a reaction is initiated with 0.40 mole of cl2 and 0.40 mole of pcl3 in a 2.0 liter container, what is the equilibrium concentration of cl2 in the same system? pcl5(g) ⇄ pcl3(g) + cl2(g)
Answers: 3

Chemistry, 22.06.2019 06:10
How many moles of gas are present if p=11 atm, v=12l, t=185k?
Answers: 1

Chemistry, 22.06.2019 07:00
The variability in marine salinity between habitats does not impact the fish living there. select the best answer from the choices provided t f
Answers: 1

Chemistry, 22.06.2019 13:30
Which statements are true concerning mineral formation? check all that apply. the slower the cooling, the larger the crystals. the faster the cooling, the smaller the crystals. crystals formed from magma are smaller than crystals formed from lava. minerals can only form in solutions when the solution is heated deep underground. when a solution cools, elements and compounds leave the solution and crystallize as minerals. minerals formed from hot water solutions can form narrow channels in the surrounding rock.
Answers: 1
You know the right answer?
You have a 25.0 L cylinder of helium at a pressure of 132 atm and a temperature of 19 [infinity]C. T...
Questions


Chemistry, 09.01.2021 18:30


Social Studies, 09.01.2021 18:30



Mathematics, 09.01.2021 18:30

Biology, 09.01.2021 18:30

Mathematics, 09.01.2021 18:30

Social Studies, 09.01.2021 18:30

English, 09.01.2021 18:30


Mathematics, 09.01.2021 18:30


Mathematics, 09.01.2021 18:30



Social Studies, 09.01.2021 18:30

History, 09.01.2021 18:30

Computers and Technology, 09.01.2021 18:30

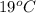 = (19 + 273) K
= (19 + 273) K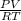
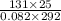
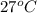 = (27 + 273) K
= (27 + 273) K