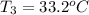Two humid streams are adiabatically mixed at 1 atm pressure to form a third stream. The first stream has a temperature of 40C, a relative humidity of 40%, and a volumetric flow rate of 3 L/s, while the second stream has a temperature of 15C, a relative humidity of 80%, and a volumetric flow rate of 1 L/s. Calculate the third stream’s temperature and relative humidity.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:00
What is the maximum amount of al2(so4)3 which could be formed from 15.84 g of al and 12.89 g of cuso4?
Answers: 2

Chemistry, 22.06.2019 05:00
Frictional forces acting on an object are often converted into energy, which causes the temperature of the object to rise slightly.
Answers: 2

Chemistry, 22.06.2019 06:00
An atom of lithium (li) and an atom of chlorine (cl) engage in a chemical reaction. which correctly describes the structure of the resulting chemical compound? hint: consider the class of each element. the chemical compound will have a network structure. the chemical compound will have triple bonds. the chemical compound will have a ball-and-stick structure. the chemical compound will have double bonds.
Answers: 2

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 3
You know the right answer?
Two humid streams are adiabatically mixed at 1 atm pressure to form a third stream. The first stream...
Questions

Mathematics, 23.04.2021 03:20


Mathematics, 23.04.2021 03:20

Mathematics, 23.04.2021 03:20

Mathematics, 23.04.2021 03:20

Mathematics, 23.04.2021 03:20

Mathematics, 23.04.2021 03:20

Mathematics, 23.04.2021 03:20

Mathematics, 23.04.2021 03:20

English, 23.04.2021 03:20

Mathematics, 23.04.2021 03:20


Physics, 23.04.2021 03:20

Mathematics, 23.04.2021 03:20


Biology, 23.04.2021 03:20

Computers and Technology, 23.04.2021 03:20

English, 23.04.2021 03:20


Mathematics, 23.04.2021 03:20

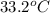 and its relative humidity is 50%.
and its relative humidity is 50%.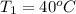 , R.H = 40%
, R.H = 40%
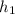 = 89 kj/kg
,
= 89 kj/kg
, 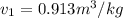
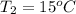
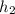 = 36.5 kj/kg
= 36.5 kj/kg
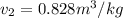
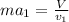
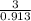
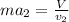
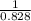
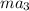 as follows.
as follows.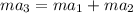
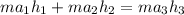
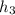 = 74.7855 kj/kg
= 74.7855 kj/kg
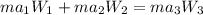
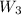 = 16.155
= 16.155
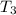 and
and 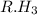 against
against