
Chemistry, 21.04.2020 20:48 25jzaldivar
The decomposition reaction 2 NOCl → 2 NO + Cl_2 has a rate law that is second order with respect to [NOCl], where k = 3.2 M^{-1}s^{-1} at a certain temperature. If the initial concentration of NOCl is 0.076 M, how many seconds will it take for [NOCl] to decrease to 0.042 M at this temperature? Do not enter units with your numerical answer.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 13:30
Why does asexual reproduction result in offspring with identicle genetic variation
Answers: 2

Chemistry, 22.06.2019 15:30
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2


Chemistry, 23.06.2019 12:30
Idid a lab for chemistry where we put nails in a copper (ii) chloride solution. 1. why did the reaction stop? which reactant was used up? how do you know? 2. describe what was happening to the atoms of iron and copper during the reaction. what is this type of reaction called? 3. what would happen to the ratio of copper to iron if you had placed more nails in the beaker? if you had let the reaction go for less time? 4. what is the accepted ratio of copper atoms to iron atoms in this reaction? account for differences between your experimental value and the accepted value. write the balanced equation for the reaction.
Answers: 2
You know the right answer?
The decomposition reaction 2 NOCl → 2 NO + Cl_2 has a rate law that is second order with respect to...
Questions


Social Studies, 30.07.2019 04:00





Mathematics, 30.07.2019 04:00


History, 30.07.2019 04:00


Biology, 30.07.2019 04:00

Mathematics, 30.07.2019 04:00



World Languages, 30.07.2019 04:00

History, 30.07.2019 04:00



Health, 30.07.2019 04:00

History, 30.07.2019 04:00

![\frac{1}{[NOCl]}=kt+\frac{1}{[NOCl]_{0}}](/tpl/images/0615/7124/81c05.png)
![[NOCl]_{0}](/tpl/images/0615/7124/069d3.png) is initial concentration of NOCl and k is rate constant.
is initial concentration of NOCl and k is rate constant.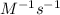 and [NOCl] = 0.042 M
and [NOCl] = 0.042 M![\frac{1}{0.042M}=[3.2M^{-1}s^{-1}\times t]+\frac{1}{0.076M}](/tpl/images/0615/7124/ee865.png)


