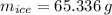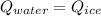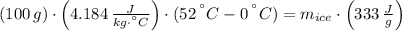Chemistry, 21.04.2020 19:22 Quanashiar
You add 100.0 g of water at 52.0 °C to 100.0 g of ice at 0.00 °C. Some of the ice melts and cools the water to 0.00 °C. When the ice and water mixture reaches thermal equilibrium at 0 °C, how much ice has melted? (The specific heat capacity of liquid water is 4.184 J/g ⋅ K. The enthalpy of fusion of ice at 0 °C is 333 J/g.) Mass of ice = g

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:30
Asap! how do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 1

Chemistry, 22.06.2019 07:00
If there is any 12 to 14 girls that need a boyfriend just follow me and let me know
Answers: 1

Chemistry, 22.06.2019 14:20
7. in the cycle, a virus integrates its dna into the host's dna, and its dna is replicated when the host dna is replicated. a. infectious b. retroviral c. lysogenic d.lytic
Answers: 1

Chemistry, 22.06.2019 18:10
Areader can tell that the meaning of “obnoxious” will include “having the quality of something” because of the .a) prefix b)pronunciation c)suffix d) word root
Answers: 3
You know the right answer?
You add 100.0 g of water at 52.0 °C to 100.0 g of ice at 0.00 °C. Some of the ice melts and cools th...
Questions





Mathematics, 21.09.2021 15:50



Advanced Placement (AP), 21.09.2021 15:50



Chemistry, 21.09.2021 15:50



Mathematics, 21.09.2021 16:00



Mathematics, 21.09.2021 16:00

Mathematics, 21.09.2021 16:00

English, 21.09.2021 16:00

Mathematics, 21.09.2021 16:00