Chemistry, 21.04.2020 18:20 Isabella1319
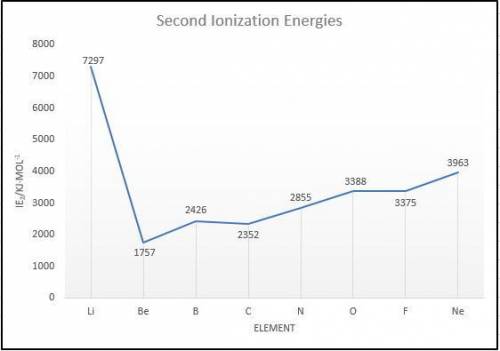
Elements in group 7A in the periodic table are called the halogens: elements in group 6A are called the chalcogens (a)
What is the most common oxidation state of the chalcogens compared to the halogens? (b) For each of the following
periodic properties. state whether the halogens or the chalcogens have larger values: atomic radii, ionic radii of the most
common oxidation state, first ionization energy. second ionization energy.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
Sally is making a model of a magnesium atom with an atomic mass number of 24 for her chemistry class. she has foam balls for the protons, neutrons, and electrons. she has added 6 neutrons to her model so far. how many more neutrons does she need to add to complete her neutral atom of magnesium?
Answers: 1



Chemistry, 23.06.2019 03:00
You have a sample of a metal, the sample is exactly 6.02 x 1023atom, if the sample has a mass 55.85 what metal is your sample made of?
Answers: 2
You know the right answer?
Elements in group 7A in the periodic table are called the halogens: elements in group 6A are called...
Questions

English, 02.05.2021 21:10

Biology, 02.05.2021 21:10


Health, 02.05.2021 21:10




Mathematics, 02.05.2021 21:10

Computers and Technology, 02.05.2021 21:10



History, 02.05.2021 21:10

Chemistry, 02.05.2021 21:10

Mathematics, 02.05.2021 21:10

Mathematics, 02.05.2021 21:10

Mathematics, 02.05.2021 21:20



Mathematics, 02.05.2021 21:20