
Chemistry, 21.04.2020 16:50 alailatrenee18541
An accident happens in the lab of Professor Utonium, and a radioactive element X is released in the form of a gas at around 4:00 am. Element X has a short half-life (25 min), and the lab would be considered safe when the concentration of X drops by a factor of 10. Considering the decomposition of element X is of first-order, what is the earliest time Professor Utonium can come back to do experiments in the lab

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
This chart represents the melting point of several substance. what besy explains the high melting point of the salt?
Answers: 2

Chemistry, 22.06.2019 00:30
Drive down any three characteristic of modern periodic table
Answers: 1

Chemistry, 22.06.2019 10:50
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3

Chemistry, 22.06.2019 12:30
The bond energy for the van der waals bond between two helium atoms is 7.9×10−4ev. assuming that the average kinetic energy of a helium atom is (3/2)kbt, at what temperature is the average kinetic energy equal to the bond energy between two helium atoms
Answers: 1
You know the right answer?
An accident happens in the lab of Professor Utonium, and a radioactive element X is released in the...
Questions




Mathematics, 09.11.2020 09:00





Business, 09.11.2020 09:00

English, 09.11.2020 09:00

English, 09.11.2020 09:00

Mathematics, 09.11.2020 09:00

English, 09.11.2020 09:00

Mathematics, 09.11.2020 09:00


Biology, 09.11.2020 09:00

Computers and Technology, 09.11.2020 09:00



Mathematics, 09.11.2020 09:00

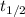 ) is 25 min. We can find the rate constant (k) using the following expression.
) is 25 min. We can find the rate constant (k) using the following expression.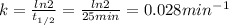
![[X]](/tpl/images/0614/8752/c2fb5.png) ) using the following expression.
) using the following expression.![[X] = [X]_0 \times e^{-k \times t}](/tpl/images/0614/8752/08bf4.png)
![[X]_0](/tpl/images/0614/8752/fbc43.png) : initial concentration of X
: initial concentration of X![\frac{[X]}{[X]_0}= e^{-k \times t}\\\frac{1/10[X]_0}{[X]_0}= e^{-0.028min^{-1} \times t}\\t=82min](/tpl/images/0614/8752/784f8.png)


