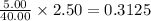A 2.50 L solution contains 5.00 g of NaOH, which has a molecular weight of 40.00
mol
Wha...


Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:50
If a substance is not at its melting or boiling point, as the heat content of a sample of matter increases, its temperature increases the number of intermolecular bonds decreases the space between particles increases the particles move faster
Answers: 2

Chemistry, 22.06.2019 01:50
Ase your answer to this question on the information below.hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant.the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen.chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product.nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1

Chemistry, 22.06.2019 10:10
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 22.06.2019 17:30
Energy defines the different "states" of matter. in no more than 3 sentences, describe the amount of kinetic energy that each of the 3 states of matter possesses and relate that to the atom/molecular motion of each "state".
Answers: 2
You know the right answer?
Questions




Health, 25.08.2019 13:00

Mathematics, 25.08.2019 13:00



World Languages, 25.08.2019 13:00

English, 25.08.2019 13:00


Social Studies, 25.08.2019 13:00

Biology, 25.08.2019 13:00

History, 25.08.2019 13:00


History, 25.08.2019 13:00

Mathematics, 25.08.2019 13:00

Chemistry, 25.08.2019 13:00


Health, 25.08.2019 13:00

Chemistry, 25.08.2019 13:00