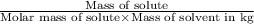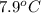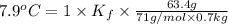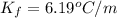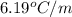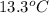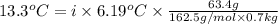Chemistry, 20.04.2020 20:50 loganrose50
When 63.4 g of glycine (C2HNO2 are dissolved in 700. g of a certain mystery liquid X, the freezing point of the solution is 7.9 °C lower than the freezing point of pure X. On the other hand, when 63.4 g of iron(III) chloride are dissolved in the same mass of X, the freezing point of the solution is 13.3 °C lower than the freezing point of pure X Calculate the van't Hoff factor for iron(III) chloride in X. Be sure your answer has a unit symbol, if necessary, and round your answer to 2 significant digits. x 10

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 11:30
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3


Chemistry, 23.06.2019 01:30
Use the periodic table to determine how many grams of oxygen would be required to react completely with 859.0 g c2h2
Answers: 3

You know the right answer?
When 63.4 g of glycine (C2HNO2 are dissolved in 700. g of a certain mystery liquid X, the freezing p...
Questions

Mathematics, 25.03.2020 16:53

Business, 25.03.2020 16:53

Chemistry, 25.03.2020 16:53

History, 25.03.2020 16:53

Biology, 25.03.2020 16:53

Business, 25.03.2020 16:53


Business, 25.03.2020 16:53



English, 25.03.2020 16:53

English, 25.03.2020 16:53


Mathematics, 25.03.2020 16:53

Mathematics, 25.03.2020 16:53


Mathematics, 25.03.2020 16:53

Mathematics, 25.03.2020 16:53

Mathematics, 25.03.2020 16:53

Mathematics, 25.03.2020 16:53

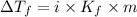
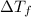 =depression in freezing point =
=depression in freezing point = 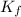 = freezing point constant
= freezing point constant