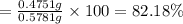Chemistry, 20.04.2020 20:32 momneedshelphmwk
A 0.5781 g sample of a pure soluble bromide compound is dissolved in water, and all of the bromide ion is precipitated as AgBr by the addition of an excess of silver nitrate. The mass of the resulting AgBr is found to be 1.1166 g. What is the mass percentage of bromine in the original compound? %

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 22.06.2019 21:00
Rays from the sun are not considered matter true or false
Answers: 2

Chemistry, 22.06.2019 23:00
What is the average rate of the reaction between 10 and 20 s?
Answers: 1
You know the right answer?
A 0.5781 g sample of a pure soluble bromide compound is dissolved in water, and all of the bromide i...
Questions

English, 16.10.2020 17:01

Chemistry, 16.10.2020 17:01



Mathematics, 16.10.2020 17:01




Computers and Technology, 16.10.2020 17:01


Mathematics, 16.10.2020 17:01

Mathematics, 16.10.2020 17:01

Mathematics, 16.10.2020 17:01



Chemistry, 16.10.2020 17:01

Mathematics, 16.10.2020 17:01


Social Studies, 16.10.2020 17:01

Biology, 16.10.2020 17:01

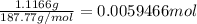
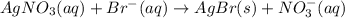
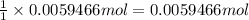 of bromide ions
of bromide ions