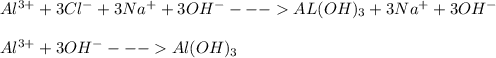Chemistry, 19.04.2020 06:26 TGJuuzouSuzuya
Write the balanced NET ionic equation for the reaction when AlCl₃ and NaOH are mixed in aqueous solution. If no reaction occurs, write only NR.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Sylvanite is a mineral that contains 28.0% gold by mass. how much sylvanite would you need to dig up to obtain 77.0 g of gold? explain how you got your answer and the steps you took. you
Answers: 3

Chemistry, 22.06.2019 11:00
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1

Chemistry, 23.06.2019 11:30
How do you calculate the mass of a product when the amounts of more than one reactant are given?
Answers: 3

Chemistry, 23.06.2019 15:30
In most resting cells, the concentration of sodium ions is higher outside of cells compared with the intracellular fluid. when cells are stimulated, sodium ion channels open, and sodium diffuses from the outside of the cell to the inside of the cell. sodium ion concentrations in a resting cell are an example of and sodium ion movement in a stimulated cell is an example of in most resting cells, the concentration of sodium ions is higher outside of cells compared with the intracellular fluid. when cells are stimulated, sodium ion channels open, and sodium diffuses from the outside of the cell to the inside of the cell. sodium ion concentrations in a resting cell are an example of and sodium ion movement in a stimulated cell is an example of potential energy; kinetic energy kinetic energy; potential energy the energy of motion; stored energy chemical work; energy stored in chemical bonds
Answers: 2
You know the right answer?
Write the balanced NET ionic equation for the reaction when AlCl₃ and NaOH are mixed in aqueous solu...
Questions

Mathematics, 02.12.2019 13:31



Mathematics, 02.12.2019 13:31



Mathematics, 02.12.2019 13:31

Mathematics, 02.12.2019 13:31





Health, 02.12.2019 13:31

English, 02.12.2019 13:31

Mathematics, 02.12.2019 13:31

Biology, 02.12.2019 13:31

Business, 02.12.2019 13:31

English, 02.12.2019 14:31

English, 02.12.2019 14:31

English, 02.12.2019 14:31