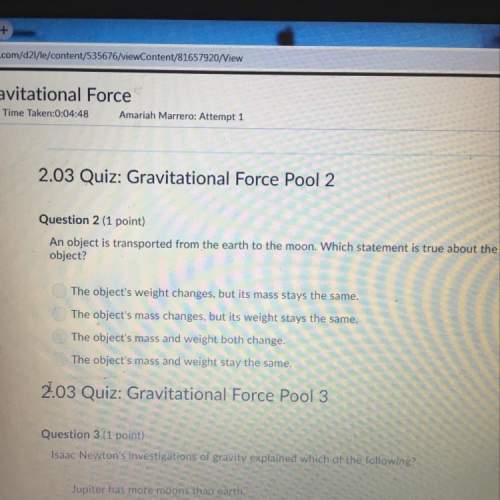
Chemistry, 17.04.2020 04:19 Mathmatician22
Consider the following reaction, equilibrium concentrations, and equilibrium constant at a particular temperature. Determine the equilibrium concentration of CO2(g). NH2COONH4(s) ↔ 2 NH3(g) + CO2(g) Kc = 1.58 × 10-8 [NH3]eq = 2.1 × 10-3 M

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 01:30
Agas is contained in a thick walled balloon when the pressure changes from 1.21 atm to 2.52 the volume changes from 3.75 l to 1.72 l and the temperature change from 293k to blank k
Answers: 3

Chemistry, 22.06.2019 05:30
Modern weaponry has increased the number of deaths in wars and violent conflicts.
Answers: 3

Chemistry, 22.06.2019 06:30
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1
You know the right answer?
Consider the following reaction, equilibrium concentrations, and equilibrium constant at a particula...
Questions











Mathematics, 25.07.2020 19:01