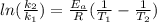The rate constant for a first order reaction is 0.060s-1. when the temperature is increased from 298K to 331 K , the rate constant increases to 0.18s-1. Calculate the activation energy for this reaction. Use kJ/mol for the units. The activation energy in kJ/mol equals (include the units in your answer):

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al
Answers: 1


Chemistry, 22.06.2019 10:20
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3

Chemistry, 22.06.2019 16:50
Answer asap need by wednesday morning calculate the ph of 0.036m naoh best answer will be brainliest
Answers: 3
You know the right answer?
The rate constant for a first order reaction is 0.060s-1. when the temperature is increased from 298...
Questions


History, 26.09.2019 14:00

Mathematics, 26.09.2019 14:00



Chemistry, 26.09.2019 14:00

Social Studies, 26.09.2019 14:00

Business, 26.09.2019 14:00

History, 26.09.2019 14:00

English, 26.09.2019 14:00

Mathematics, 26.09.2019 14:00

Biology, 26.09.2019 14:00

Social Studies, 26.09.2019 14:00

Mathematics, 26.09.2019 14:00



Social Studies, 26.09.2019 14:00

Social Studies, 26.09.2019 14:00


Mathematics, 26.09.2019 14:00