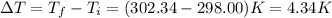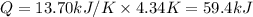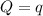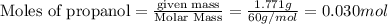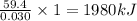Chemistry, 16.04.2020 01:46 maggie123456751
The combustion of 1.771 g of propanol ( C 3 H 7 OH ) increases the temperature of a bomb calorimeter from 298.00 K to 302.34 K . The heat capacity of the bomb calorimeter is 13.70 kJ/K . Determine Δ H for the combustion of propanol to carbon dioxide gas and liquid water.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1

Chemistry, 22.06.2019 15:30
Which of the following are correct values for the ideal gas laws constant r
Answers: 1

Chemistry, 22.06.2019 16:50
Which of the following is an indication that a substance has undergone a chemical change? a. no new product has been formed. b. the color of the substance has not changed. c. the original constitute has not changed. d. the molecular structure has changed.
Answers: 1

Chemistry, 23.06.2019 01:00
Which polymers are most closely related? a. protein and nucleic acids b. cellulose and starch c. nucleic acids and starch d. nucleic acids and cellulose
Answers: 2
You know the right answer?
The combustion of 1.771 g of propanol ( C 3 H 7 OH ) increases the temperature of a bomb calorimeter...
Questions

Biology, 23.04.2020 22:20

Geography, 23.04.2020 22:20


Mathematics, 23.04.2020 22:20

History, 23.04.2020 22:20


Mathematics, 23.04.2020 22:20



History, 23.04.2020 22:20





Mathematics, 23.04.2020 22:21

Business, 23.04.2020 22:21


Business, 23.04.2020 22:21


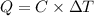
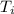 = 298.00 K
= 298.00 K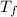 = 302.34 K
= 302.34 K