
Suppose you add 0.2469 g of PbCl2(s) to 50.0 mL of water. In the resulting saturated solution, you find that the concentration of PbX2+(aq) is 0.0159 M and the concentration of C l − ( a q ) ClX−(aq) is 0.0318 M. What is the value of the equilibrium constant, Ksp, for the dissolution of PbCl2?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 07:20
After watching the video "zinc strip in copper nitrate solution", and reading the instructions, click on the link labeled "start" just below the drawing of the pencil tip. follow the direction to complete the 3x3 grid. answer the below questions for the portion of the activity in which sn(s) is placed in agno3(aq)
Answers: 1

Chemistry, 22.06.2019 17:00
In a heat engine of 1000 j of heat enters the system and the piston does 500 j of work what is the final internal energy of the system if the inital energy was 2000 j we have to do all of these down here 1)write the equation 2)list out your know variables 3)plug the numbers into the equations 4)solve 5)write your solution statemtn that includes inital energuy and final energuy added
Answers: 1

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium
Answers: 1
You know the right answer?
Suppose you add 0.2469 g of PbCl2(s) to 50.0 mL of water. In the resulting saturated solution, you f...
Questions

Biology, 17.04.2020 23:08


English, 17.04.2020 23:08


Mathematics, 17.04.2020 23:08

History, 17.04.2020 23:08



Mathematics, 17.04.2020 23:08

Health, 17.04.2020 23:08


Mathematics, 17.04.2020 23:08



Mathematics, 17.04.2020 23:08


Mathematics, 17.04.2020 23:08


Mathematics, 17.04.2020 23:08

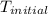 = a - -
= a - -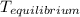 = a - s s 2s
= a - s s 2s

