Chemistry, 16.04.2020 00:23 lerasteidl
Measurements show that the enthalpy of a mixture of gaseous reactants increases by 243.kJ during a certain chemical reaction, which is carried out at a constant pressure. Furthermore, by carefully monitoring the volume change it is determined that −174.kJ of work is done on the mixture during the reaction. Calculate the change of energy of the gas mixture during the reaction in kJ.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:10
Answer from each drop-down menu. e characteristics of a borane molecule (bh). the lewis structure and table of electronegativities are given olecular shape is and the molecule is reset next erved. search e a
Answers: 2

Chemistry, 22.06.2019 13:30
How many protons, electrons, and neutrons are in each of the following isotopes? a. zirconium-90 b. palladium-108 c. bromine-81 d. antimony-123
Answers: 1

Chemistry, 22.06.2019 18:30
Which of the following nuclei would be the least stable a 2 protons, 2 neutrons b 1 proton 1 neutron c 1 proton 3 neutrons d 1 proton 2 neutrons
Answers: 3

Chemistry, 23.06.2019 02:00
When an experimenter draws a conclusion that he assumes will apply to all situations set up similarly to his test situation, even though he cannot possibly have examined all possible test scenarios, the experimenter is using deductive reasoning inductive reasoning abductive reasoning subjective reasoning
Answers: 1
You know the right answer?
Measurements show that the enthalpy of a mixture of gaseous reactants increases by 243.kJ during a c...
Questions

Chemistry, 11.12.2020 02:00

English, 11.12.2020 02:00

Mathematics, 11.12.2020 02:00

History, 11.12.2020 02:00

Mathematics, 11.12.2020 02:00


Mathematics, 11.12.2020 02:00




Mathematics, 11.12.2020 02:00




English, 11.12.2020 02:00

Computers and Technology, 11.12.2020 02:00

English, 11.12.2020 02:00

Mathematics, 11.12.2020 02:00


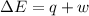
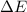 =Change in internal energy
=Change in internal energy