
Chemistry, 15.04.2020 17:29 jessemartinez1
How many of the following molecules are polar? Pcl5 cos xeo3 sebr2

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:10
Answer from each drop-down menu. e characteristics of a borane molecule (bh). the lewis structure and table of electronegativities are given olecular shape is and the molecule is reset next erved. search e a
Answers: 2

Chemistry, 22.06.2019 09:20
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3

Chemistry, 22.06.2019 11:50
Which of the following statements about hybrid orbitals is or are true? choose all that apply. choose all that apply. under sp2 hybridization, the large lobes point to the vertices of an equilateral triangle. after an atom undergoes sp hybridization there is one unhybridized p orbital on the atom. the angle between the large lobes of sp3 hybrids is 109.5∘
Answers: 2

You know the right answer?
How many of the following molecules are polar? Pcl5 cos xeo3 sebr2...
Questions



Chemistry, 06.05.2020 04:12


Geography, 06.05.2020 04:12

Computers and Technology, 06.05.2020 04:12





Business, 06.05.2020 04:12





English, 06.05.2020 04:12

English, 06.05.2020 04:12

SAT, 06.05.2020 04:12


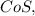
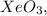
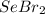 .
.

