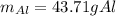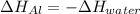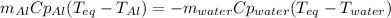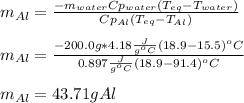Chemistry, 15.04.2020 15:49 ljcervantes4824
A chunk of aluminum at 91.4°C was added to 200.0 g of water at 15.5°C. The specific heat of aluminum is 0.897 J/g°C, and the specific heat of water is 4.18 J/g°C. When the temperature stabilized, the temperature of the mixture was 18.9°C. Assuming no heat was lost to the surroundings, what was the mass of aluminum added?
A. 243 g
B. 34.7 g
C. 41.7 g
D. 43.7 g

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:30
In pea plants, the allele for tallness (t) is dominant to the allele for shortness (t). in the cross between a tall pea plant and a short pea plant shown below, what is the probability that the resulting offspring will be tall? whats the percent
Answers: 1


Chemistry, 22.06.2019 13:00
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3
You know the right answer?
A chunk of aluminum at 91.4°C was added to 200.0 g of water at 15.5°C. The specific heat of aluminum...
Questions

Physics, 07.11.2020 01:00

Physics, 07.11.2020 01:00


Arts, 07.11.2020 01:00

Mathematics, 07.11.2020 01:00

Biology, 07.11.2020 01:00


Mathematics, 07.11.2020 01:00


Mathematics, 07.11.2020 01:00




English, 07.11.2020 01:00


Computers and Technology, 07.11.2020 01:00

Biology, 07.11.2020 01:00


English, 07.11.2020 01:00

Social Studies, 07.11.2020 01:00