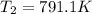Chemistry, 15.04.2020 02:57 christiannpettyy
The compound 1,1-difluoroethane decomposes at elevated temperatures to give fluoroethylene and hydrogen fluoride: CH3CHF2(g) → CH2CHF(g) + HF(g) At 460 °C, k = 5.8 × 10-6 s-1 and Ea = 265 kJ/mol. To what temperature (in K) would you have to raise the reaction to make it go four times as fast?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:20
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 09:00
Astudent is asked to identify and element that is pale yellow brittle solid and does not conduct electricity. at which location in this periodic table would the element most likely be found?
Answers: 2

Chemistry, 22.06.2019 10:10
Stage in which a star’s outer layers have started to cool and grow outward?
Answers: 3

Chemistry, 22.06.2019 11:00
Ais a mountain created from eruptions of lava, ash, rocks, and hot gases.
Answers: 1
You know the right answer?
The compound 1,1-difluoroethane decomposes at elevated temperatures to give fluoroethylene and hydro...
Questions


Mathematics, 11.06.2020 14:57

Mathematics, 11.06.2020 14:57

Mathematics, 11.06.2020 14:57



Mathematics, 11.06.2020 14:57


Geography, 11.06.2020 14:57


Biology, 11.06.2020 14:57


Mathematics, 11.06.2020 14:57






Mathematics, 11.06.2020 14:57

Mathematics, 11.06.2020 14:57

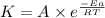
![\log (\frac{K_2}{K_1})=\frac{Ea}{2.303\times R}[\frac{1}{T_1}-\frac{1}{T_2}]](/tpl/images/0600/9306/6d953.png)
 = rate constant at
= rate constant at  =
= 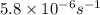
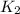 = rate constant at
= rate constant at 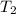 =
= 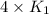
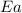 = activation energy for the reaction = 265 kJ/mol = 265000 J/mol
= activation energy for the reaction = 265 kJ/mol = 265000 J/mol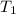 = initial temperature =
= initial temperature = 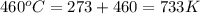
![\log (\frac{4\times K_1}{K_1})=\frac{265000J/mol}{2.303\times 8.314J/mole.K}[\frac{1}{733K}-\frac{1}{T_2}]](/tpl/images/0600/9306/ddf14.png)