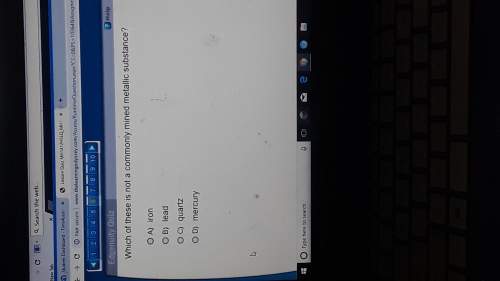
In a redox reaction, why does an element's oxidation number decrease?
O A. An element's oxidat...

Chemistry, 15.04.2020 00:24 amiechap12
In a redox reaction, why does an element's oxidation number decrease?
O A. An element's oxidation number decreases because it gained electrons.
B.
An element's oxidation number decreases because it exchanged electrons.
• C.
An element's oxidation number decreases because it lost electrons.
OD. An element's oxidation number decreases because it was heated.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:30
What is the relation between concentration of reactants and the rate of chemical reaction?
Answers: 1

Chemistry, 22.06.2019 05:00
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

Chemistry, 22.06.2019 05:50
Calculate the number of molecules present in 0.750 mol of mgo.
Answers: 3

You know the right answer?
Questions

Mathematics, 03.11.2020 04:30

Mathematics, 03.11.2020 04:30

Advanced Placement (AP), 03.11.2020 04:30

Physics, 03.11.2020 04:30


Mathematics, 03.11.2020 04:30


Biology, 03.11.2020 04:30

Business, 03.11.2020 04:30

Mathematics, 03.11.2020 04:30

Geography, 03.11.2020 04:30


History, 03.11.2020 04:30

History, 03.11.2020 04:30

Social Studies, 03.11.2020 04:30

English, 03.11.2020 04:30

Mathematics, 03.11.2020 04:30

History, 03.11.2020 04:30

Biology, 03.11.2020 04:30

Mathematics, 03.11.2020 04:30